Fermention Expertise
Innovating Fermentation Processes for Seamless Development and Manufacturing
Curia’s dedicated fermentation teams combines state-of-the-art facilities with expert knowledge to deliver customized solutions, from development to manufacturing. Our track record showcases unparalleled quality and regulatory compliance. With a multidisciplinary approach and versatile feedback control systems, we are adept at tackling challenges from benchtop experiments to full-scale production.
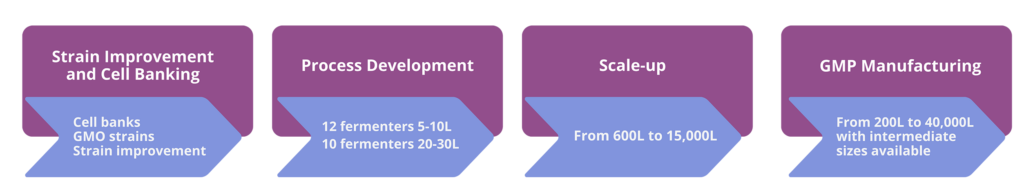
- Microbial Strain Improvement: Conducted in cutting-edge labs with precision fermentation techniques, Curia is able to work with bacteria, yeast and fungi (both engineered and natural strains
- Process Optimization: Scale-up capabilities for preclinical, clinical trials or manufacturing
- Proven Success: A variety of case studies highlight Curia’s ability to rapidly deliver robust, cost-advantaged processes, new intellectual property (IP), and novel natural products (microbial collection)
Flexible Scale, Customization and Downstream Capabilities
With over 20 years of experience, Curia excels in manufacturing, isolating and purifying complex biological mixtures. Our infrastructure includes reactors ranging from 2L to 7,000L with advanced monitoring tools and broad DSP capabilities.
Key Capabilities Include:

Industrial Fermentation and Biotransformation
Curia excels in:
- Seamlessly integrating biocatalysis with advanced chemical synthesis
- Creating innovative and efficient processes that optimize both safety and scalability
- Multidisciplinary approaches to unlock new intellectual property opportunities
- Downstream process excellence, including scale-up capabilities for large volumes with solvent-optimized processes
Your Product, Our Expertise
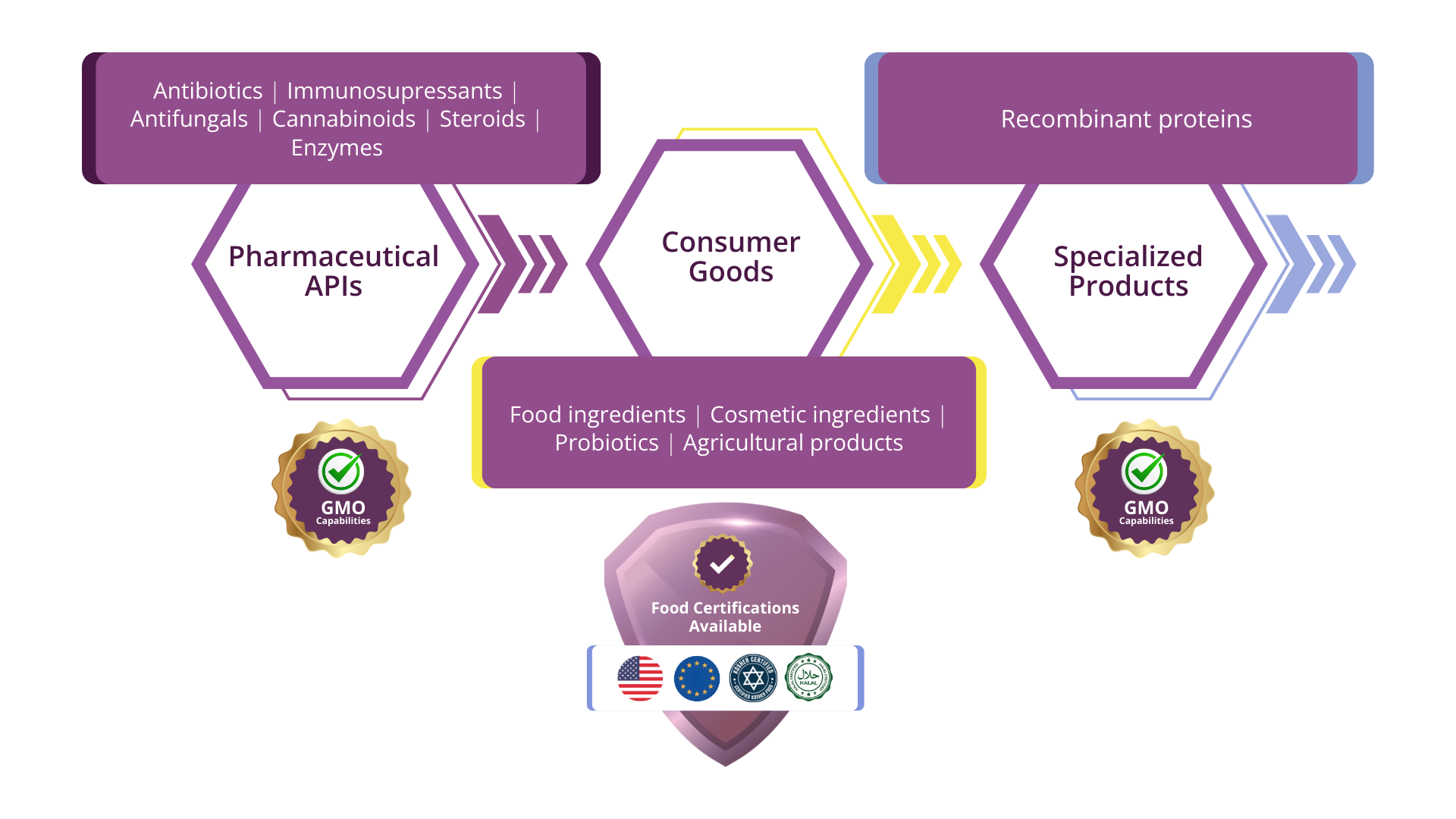
Curia Network
Fermentation Insights
Frequently Asked Questions
What is microbial fermentation?
Microbial fermentation is a metabolic process that uses microorganisms, such as bacteria, yeast, or fungi to convert
raw materials (carbon sources like glucose) into high-value products. These “cellular factories” can produce proteins,
enzymes, small molecules, and primary or secondary metabolites. Unlike chemical synthesis, it relies on the biological
machinery of the cell to build complex molecules.
Where is fermentation used today?
Beyond its ancient roots in food and beverage, industrial fermentation is now the backbone of several high-tech
sectors, including but not limited to:
- Pharmaceuticals: Production of recombinant proteins (e.g., insulin), vaccines, antibiotics, and plasmid DNA for gene therapy.
- Specialty Chemicals: Organic acids, amino acids, and high-purity solvents.
- Nutraceuticals: Vitamins (B12, Riboflavin), omega-3 fatty acids, and probiotics.
- Agriculture: Bio-pesticides and growth stimulants.
What’s the difference between fermentation and biocatalysis?
While both involve biology, they are distinct operational strategies:
- Fermentation (De Novo Synthesis): The microorganism is alive and growing. It consumes a substrate (sugar) and uses its entire metabolic pathway to build a product from scratch. It is recommended for complex molecules that require many steps to build.
- Biocatalysis (Biotransformation): This uses specific enzymes (either isolated or within a “resting” cell) as a catalyst for a specific chemical reaction. You start with a precursor molecule and “transform” it into the final product. A common use case is achieving extreme precision (e.g., specific chirality) in a chemical synthesis route.
How do these technologies contribute to “Green Chemistry”?
Fermentation and biocatalysis are pillars of sustainable manufacturing. They offer:
- Mild Conditions: Reactions occur at ambient temperature and pressure in aqueous (water-based) environments, drastically reducing energy consumption compared to high-heat chemical reactors.
- Waste Reduction: Biocatalysts are highly selective, meaning fewer side reactions and less toxic byproduct waste.
- Step Economy: Biocatalysis can often combine three or four traditional chemical steps into a single “one-pot” enzymatic reaction.
What are the primary regulatory hurdles?
Transitioning to biological processes introduces unique compliance requirements:
- Strain Characterization: Regulators (FDA/EMA) require data on the genetic stability of the production strain to ensure consistent product quality across batches.
- Impurity Profiling: Removing “host cell proteins” (HCPs) and DNA to parts-per-million levels is critical, particularly for injectable drugs.
- Process Validation: Biological systems are “living” and variable, requiring rigorous “Quality by Design” (QbD) to control parameters like dissolved oxygen and nutrient feed rates.
- Regulatory Refiling: Switching an existing chemical process to a bio-based one often requires a “Type II” variation or a new filing, which can delay market implementation.
What is the current market size for these services?
The market is currently experiencing “hyper-growth” driven by the biologics boom. It is currently valued at
approximately $4.5 – $5.2 billion (2024/2025) and projected to reach over $12 billion by 2033 (12.5% CAGR). Moreover,
industrial enzymes (biocatalysis) are growing at ~6.4% CAGR, expected to hit nearly $1 billion by 2030. There is a clear
outsourcing trend. Over 44% of manufacturing is now handled by CDMOs as companies seek to avoid the high CAPEX
of building their own bioreactor suites.
What trends are shaping the future?
- Precision Fermentation: Using CRISPR to program microbes for animal-free proteins (e.g., collagen, dairy).
- Continuous Manufacturing: Switching from “batch” to continuous flow to increase yield and reduce footprint.
- AI & Digital Twins: Using machine learning to predict large-scale (10,000L+) performance before leaving the lab.
- Cell-Free Synthesis: Eliminating the cell entirely to use enzymatic “machinery” in a soup, bypassing toxicity limits of living organisms.
Why choose a CDMO for these services?
Scaling from a 2L benchtop to a 5,000L+ industrial fermenter is capital-intensive and technically risky. Curia
provides the specialized scale-up expertise, GMP compliance, and downstream purification (DSP) infrastructure that
most companies cannot maintain in-house.



