Supporting you from early stage development to commercial manufacturing, our proven track record with parenteral formulations demonstrates our ability to be your partner for the entire product life-cycle.
What sets Curia apart?
Curia’s network of drug product sites apply a bespoke approach with an emphasis on quality, safety, and efficacy, ensuring reliable product as you move from development to commercial manufacturing
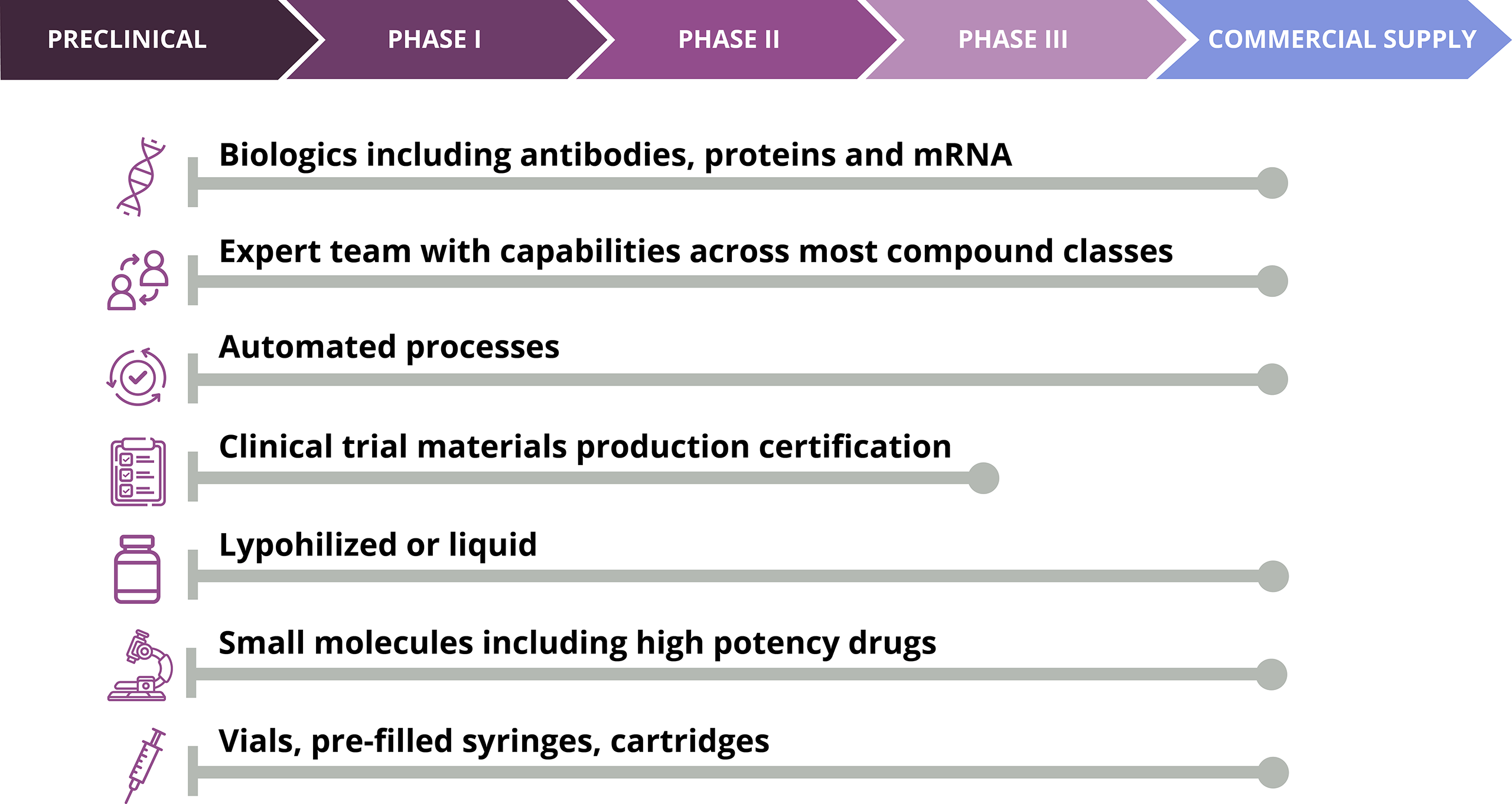
- End-to-end development in sterile fill-finish
- Lyophilization expertise in all phases
- Formulation and process development support
- Capability and capacity in vials, pre-filled syringe, and cartridges
- Handling of Controlled drugs (Schedule I to V)
- Handling of HPAPI, Cyto/non-Cyto Small Molecule, and Biologics up to BSL2
- Highly efficient and reliable internal/external technology transfer process
DRUG PRODUCT Development
Embracing complexity worldwide
Curia works with complex APIs, including generics, sterile APIs, potent and cytotoxic compounds, and controlled substances – from clinical to commercial scale – at advanced facilities around the world.
Modality agnostic, first principle formulation development designed around target product profile. Curia experts support the development of solutions, suspensions, emulsions, lipid nanoparticles, and lyophilized dosage forms. Addressing inherent complexities to provide robust, scalable products and processes for cGMP drug product manufacture.
DRUG PRODUCT MANUFACTURING
Various formats for when you need them
Curia has the demonstrated ability to provide start-to-finish sterile fill-finish in various formats, including vials, pre-filled syringes, and cartridges, supporting your program from preclinical through commercialization
Our Approach is a Process, not a Platform
At each product development milestone, you can easily transfer into Curia to advance and streamline the process.
Our Target Product Profile (TPP) covers:
- Route of administration
- Dosage concentration
- Container closure system
- Critical Quality Attributes
- Critical Process Parameters
Sterile Injectables
Our expertise lies in highly viscous formulations along with manufacturing complex injectables, lipid nanoparticles (LNP), highly potent and cytotoxic compounds, and controlled substances. We embrace complexity.
The Power Of Our Drug Product Network
Save time, cut costs, and seamlessly advance your product with our extensive network of sites and experts in aseptic processing. With Curia, you have a customized experience with a well-versed, dedicated program manager and team of subject matter experts from development through commercialization.