Sterile API Manufacturing
Curia provides sterile APIs for over 300 clients in more than 30 countries around the world. Our customers trust us because of our excellent track record of regulatory compliance and quality assurance.
There are several different methods which can be used to sterilize APIs, such as the terminal ones that are heat (dry or wet) and radiation. Curia offers a technology that is used in situations where the terminal ones do not guarantee the stability of the API, neither a good impurity profile. This is the aseptic processing, working under closed systems design (as isolators) throughout the entire process to ensure sterility and to minimize the risk of contamination.
Curia is uniquely positioned to handle the most complicated challenges of manufacturing
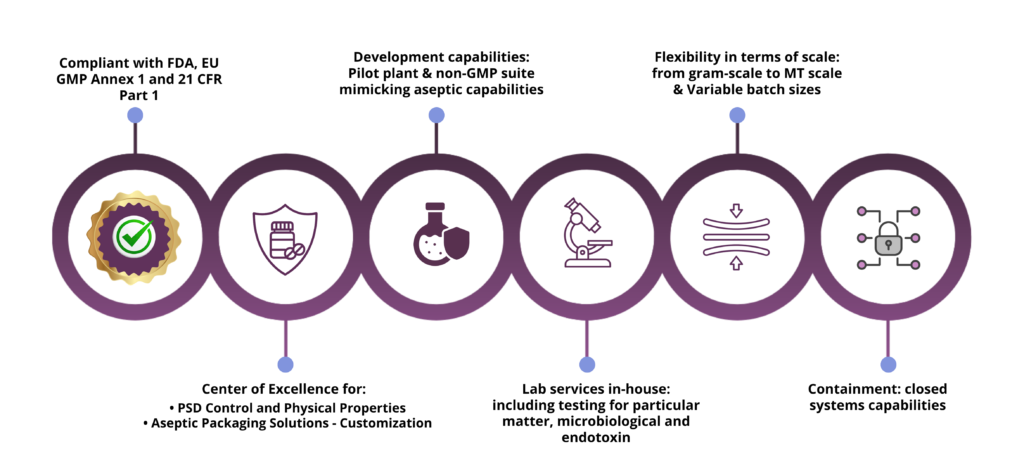
With over 20 years of experience, Curia is here to help you identifying best conditions and parameters to perform Aseptic API sterilizing filtration, with three different manufacturing suites available, and following a four-step strategy for successful sterile API manufacturing:
Stage One: Dissolution and Sterile Filtration
Stage Two: Crystallization
Stage Three: Filtration and Drying
Stage Four: Physical Treatment and Packaging
Apart from the main process, we have secondary processes that we use to minimize risk in sterile API manufacturing. These include operator training to prevent contamination during handling, environmental monitoring to check all conditions and operations are right to assure the safety conditions, pressure cascade control to avoid contamination between rooms and particle control strategies to avoid foreign particles.





